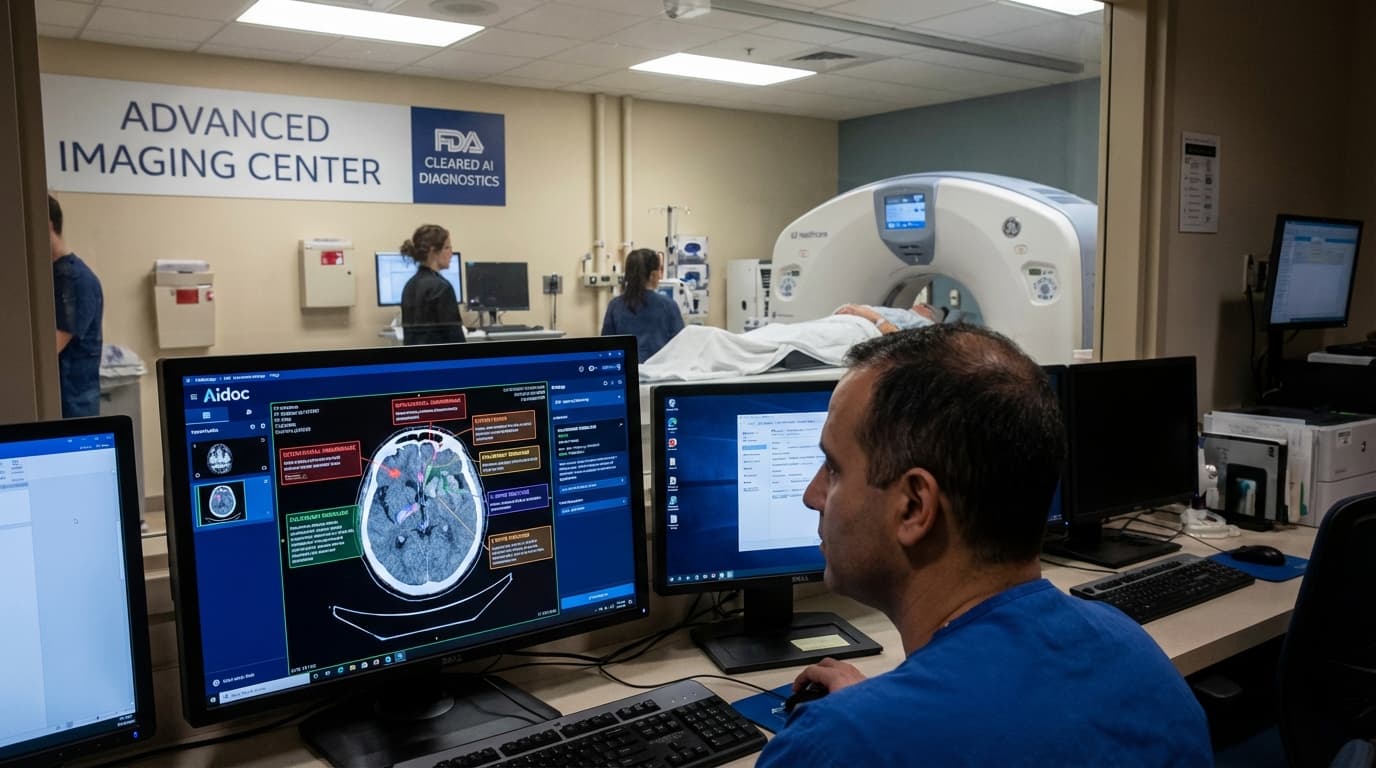
FDA Approves Aidoc’s AI Tool for Detecting Multiple Conditions from CT Scans
Aidoc’s newly cleared AI tool represents a promising advancement in radiological diagnostics by enabling the detection of multiple conditions simultaneously from computed tomography scans. This innovation could streamline diagnosis, improve patient outcomes, and reduce diagnostic errors in clinical settings.
In a move that could significantly impact the field of medical imaging and diagnostics, the U.S. Food and Drug Administration (FDA) has granted clearance to Aidoc’s artificial intelligence (AI) tool designed to analyze computed tomography (CT) scans and detect 14 different medical conditions from a single scan. This development embodies a pivotal advancement in the integration of AI technologies within clinical radiology, promising to enhance diagnostic efficiency, accuracy, and patient care.
The AI-powered system from Aidoc leverages sophisticated machine learning algorithms to evaluate CT scan images and identify various potential pathological findings that may otherwise require multiple tests or detailed manual interpretation by radiologists. With this technology, a single CT scan can be rapidly analyzed to detect a wide array of conditions, which is instrumental in accelerating the clinical decision-making process.
The FDA clearance underscores that Aidoc’s AI tool meets rigorous safety and effectiveness standards, ensuring it can reliably assist healthcare professionals in clinical environments. This is particularly relevant given the increasing volume of imaging studies that radiologists must interpret and the ongoing demand for timely and accurate diagnostic information.
Aidoc’s technology is capable of recognizing a diverse set of conditions visible on CT imaging, potentially ranging from acute anomalies such as hemorrhages or ischemic changes to findings related to trauma, vascular issues, infections, or other diseases. By automating the initial detection and characterization phases, the AI tool can prioritize cases that warrant immediate attention, enabling more efficient allocation of radiological and clinical resources.
Moreover, the integration of such AI tools into radiology workflows could reduce variability in image interpretation and help mitigate human error. This aspect is vital for improving diagnostic consistency and helping to ensure that critical findings are not overlooked, particularly in busy hospital settings or emergency departments.
The adoption of Aidoc’s AI tool is anticipated to have broader implications for patient outcomes, including faster diagnosis, earlier initiation of appropriate treatments, and possibly reductions in unnecessary follow-up imaging or procedures. As medical imaging continues to evolve with the incorporation of AI, regulatory clearances like this represent essential milestones that facilitate the transition from experimental applications to routine clinical use.
Notably, this FDA approval comes amidst a growing landscape where artificial intelligence is becoming instrumental across various healthcare domains, especially in medical diagnostics. The ability to simultaneously detect multiple conditions from the same imaging study reflects the potential of AI to enhance multidimensional clinical data analysis and foster more holistic patient evaluations.
Healthcare institutions and providers planning to integrate AI systems like Aidoc’s must also consider the operational, ethical, and training considerations associated with AI-driven diagnostics. Ensuring that clinical staff are adequately trained to collaborate with AI tools and that workflows are optimized for seamless AI integration remains a critical aspect of translating technological advancements into actual clinical benefits.
In summary, the FDA’s clearance of Aidoc’s AI tool signifies a critical step in the advancement of medical imaging technologies, marking progress toward more comprehensive, rapid, and accurate diagnostic processes through artificial intelligence. This milestone paves the way for expanded use of AI in radiology and potentially other diagnostic imaging fields, driving forward the vision of AI-augmented medical practice.
Source: STAT+ FDA clears Aidoc tool to detect 14 different conditions from a CT scan
Join the BioIntel newsletter
Get curated biotech intelligence across AI, industry, innovation, investment, medtech, and policy—delivered to your inbox.